In such cases, the charge of the metal ion is included as a Roman numeral in parentheses immediately following the metal name. 
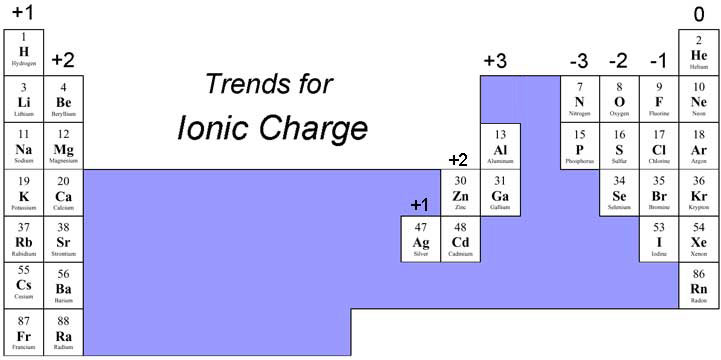
For example, in a binary ionic compound of iron and chlorine, iron typically exhibits a charge of either 2+ or 3+, and the two corresponding compound formulas are FeCl 2 and FeCl 3. The charge of a metal ion is determined from the formula of the compound and the charge of anion. Compounds of these metals with nonmetals are named with the same method as binary compounds, except the charge of the metal ion is specified by a Roman numeral in parentheses after the name of the metal. Most of the transition metals and some main group metals can form two or more cations with different charges. For example, the name for CaSO 4 is calcium sulfate. For example, the name for Na 2O is sodium oxide.Ĭompounds containing polyatomic ions are also named similarly to those containing only monatomic ions, i.e., by naming first the cation and then the anion. The name of a binary compound containing monatomic ions consists of the name of the cation (the name of the metal) followed by the name of the anion (the name of the nonmetallic element with its ending replaced by the suffix -ide). For example, the ionic compound sodium oxalate is composed of Na + and C 2O 4 2− ions combined in a 2:1 ratio, and its formula is written as Na 2C 2O 4. However, the formulas for some ionic compounds containing polyatomic ions are not empirical formulas. For compounds containing only monatomic ions (such as NaCl) and for many compounds containing polyatomic ions (such as CaSO 4), these formulas are just the empirical formulas. Ionic compounds are symbolized by formula, indicating the relative numbers of its constituent ions. The compound is electrically neutral, and its formula shows a total count of three Ca, two P, and eight O atoms. This formula indicates that there are three Ca 2+ ions (total six positive charges) for every two PO 4 3− groups (total six negative charges). The compound contains the polyatomic ion PO 4 3−, consisting of one phosphorus atom and four oxygen atoms, and having an overall charge of 3−. For example, the formula for calcium phosphate, one of the minerals in our bones, is Ca 3(PO 4) 2. Parentheses are used in the formula to indicate polyatomic ions that behave as a unit. As with simple ionic compounds, these compounds must also be electrically neutral, so their formulas can be predicted by treating the polyatomic ions as discrete units. Polyatomic ions are a group of bonded atoms that act as discrete units, carrying an overall charge. Many ionic compounds contain polyatomic ions as the cation, the anion, or both. Thus, the compound will be electrically neutral, with the same number of positive and negative charges. Two aluminum ions, each with a charge of 3+, would give us six positive charges, and three oxide ions, each with a charge of 2−, would give us six negative charges. The formula of an ionic compound must have a ratio of ions such that the numbers of positive and negative charges are equal.įor example, if a compound contains aluminum and oxygen in the form of Al 3+ and O 2−, the formula of the compound would be Al 2O 3. Thus, ionic compounds are electrically neutral, even though they contain positive and negative ions. In every ionic compound, the total number of positive charges of the cations equals the total number of negative charges of the anions. For example, copper can form ions with a 1+ or 2+ charge, and iron can form ions with a 2+ or 3+ charge. Transition metals and some other metals often exhibit variable charges that are not predictable by their location in the table. The name of the anions is the name of the nonmetallic element with its ending replaced by the suffix -ide, so O 2− is called oxide. The anion, O 2−, has the same number of electrons as the next noble gas – neon. This results in an anion with 8 protons, 10 electrons, and a 2− charge, and is symbolized as O 2−. For example, a neutral oxygen atom, with 8 protons and 8 electrons, readily gains two electrons. That is, atoms of group 17 gain one electron and form anions with a 1− charge atoms of group 16 gain two electrons and form ions with a 2− charge, and so on. Moving from the far right to the left on the periodic table, nonmetal elements often gain electrons to form anions with the same number of electrons as an atom of the next noble gas in the periodic table, and a negative charge equal to the number of groups moved left from the noble gases. The compound formed by this transfer is stabilized by the electrostatic attractions (ionic bonds) between the oppositely charged ions. An element composed of atoms that readily lose electrons (a metal) can react with an element composed of atoms that readily gain electrons (a nonmetal) to produce ions through complete electron transfer. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
